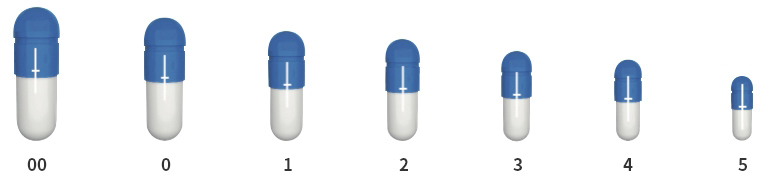
| Size (♯) | 00 | 0 | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|---|---|
| Volume (mL) | 0.93 | 0.68 | 0.49 | 0.37 | 0.28 | 0.21 | 0.13 |
Pharmaceutical capsules

High-quality capsules that have been the market standard over many years.


More than 90% pharmaceutical capsules made in Qualicaps® are imprinted to enhance distinguishability.
Two imprinting methods are available:
・Offset printing with various color ink
・UV lazer printing with no ink : Qualicaps®'s own technology
Offset printing uses edible ink,
in which various colors are available.
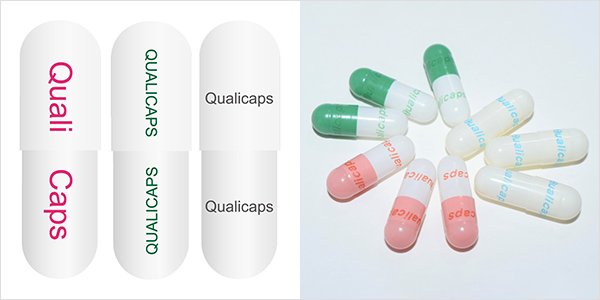
UV lazer printing technology utilizes the color change of titanium oxide by UV irradiation.
UV lazer can print fine design to capsules.
| Offset printing | UV lazer printing | |
|---|---|---|
| Color | Various | Gray |
| Fineness | Good | Excellent |
Available in 7 sizes, from 00 to 5.
In addition, please contact us for inquiries about the special size.

| Size (♯) | 00 | 0 | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|---|---|
| Volume (mL) | 0.93 | 0.68 | 0.49 | 0.37 | 0.28 | 0.21 | 0.13 |
The standard numbers of capsules packed in each case for outside of Japan are as follows.
| Capsule size (♯) | Standard quantities | |
|---|---|---|
| 00 | 70,000 | |
| 0 | 90,000 | |
| 1 | Transparent capsules | 100,000 |
| Opaque capsules | 125,000 | |
| 2 | 150,000 | |
| 3 | 200,000 | |
| 4 | 250,000 | |
| 5 | 400,000 | |
QUALI-G™ capsules have excellent dissolution properties, with not less than 80% of the capsule contents released within 5 minutes.
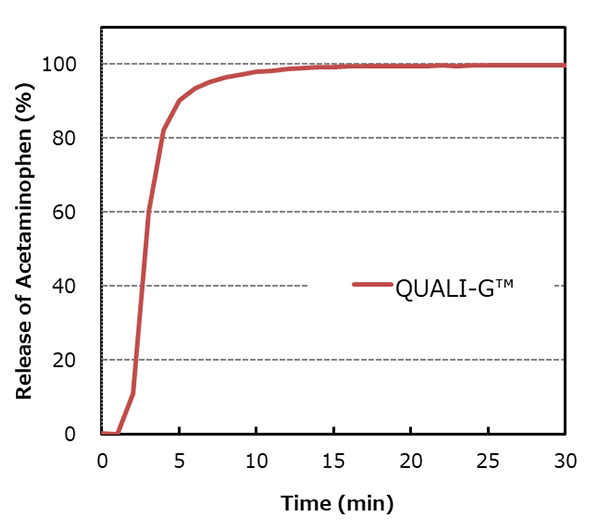
Capsule size: ♯3
Apparatus: Paddle method, 50 rpm, UV detection
Dissolution medium: water, 900 mL
Capsule fill formulation: 20% acetaminophen mixture
Fill weight: 150 mg
We prepare accreditations and/or certifications for QUALI-G™. For further reference, please contact us for further information.
| Residual Solvents Statement | BSE/TSE Statement | Elemental Impurities Statement |
| GMO Statement | Allergen Statement | Melamine Statement |
| Nitrosamines Statement | Titanium Oxide Statement | Gluten Statement |
| Irradiation Statement | ||
| SDS | Capsule Specifications | Flow of Manufacturing Capsules |
| Test Methods | Capsule Theoretical Formula | COA Sample |
Additionally, Qualicaps® received ISO9001 and ISO14001 certification.
See ″ISO Management″ for further details.